Ziling (Ben) Xue

ADDRESS
Address Lab
Phone
Ziling (Ben) Xue
Professor
Dr. Xue received his B.S. in chemistry from Nanjing University of Pharmacy-Nanjing University in 1982 and his Ph.D. in chemistry from the University of California at Los Angeles in 1989. Following postdoctoral research at Indiana University, he joined the faculty of the University of Tennessee in 1992.
Research
Our research is centered on two areas: (1) Probing magnetism of metal complexes for potential applications as single-molecule magnets (SMMs) and qubits; (2) Studying Haldane topological spin chains as new quantum materials.
(1) Probing Magnetism of Metal Complexes
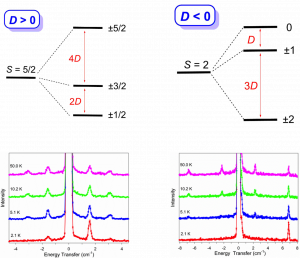
Many transition metal and lanthanide complexes have unpaired electrons, making their molecules magnetic. Recently, molecular magnetism has been actively studied as many such complexes are SMMs and qubit (quantum bit) candidates. SMMs may be used as new data storage materials with much larger capacities than the current digital media. In addition, SMMs are potential candidates for quantum computing. Challenges in SMM studies include: (a) Determination of magnetic separations in SMMs; (b) Understanding of magnetic relaxations. We have used state-of-the-art techniques such as far-IR magneto-spectroscopy (FIRMS), Raman magneto-spectroscopy (RaMS), and inelastic neutron scattering (INS) to determine the magnetic separations in SMMs and to probe magnetic relaxations. In addition, the spectroscopic studies lead to the rare observation of spin-vibrational couplings (known as spin-phonon couplings), which are critical to magnetic relaxation of the molecules from excited states. The studies are collaborations with scientists at National High Magnetic Field Laboratory, Oak Ridge National Laboratory, and NIST Center for Neutron Research, among others.
Figure 1. INS spectra of Mn(TPP) (Right; S = 5/2; D > 0) and Mn(TPP)Cl (Left; S = 2; D < 0). TPP2- = tetraphenylporphyrinate.
(2) Studying Haldane Topological Spin Chains as New Quantum Materials
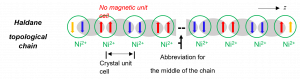
Haldane topological materials contain unique antiferromagnetic (AF) chains of, e.g., Ni2+ (S = 1) ions with one unpaired electron at each end of the chains in the ground state. The chains use ligands such as oxalate (C2O42- or ox2-) to bridge neighboring Ni2+ ions forming polymeric chains. Duncan Haldane first proposed such AF coupling in the chains as the most fundamental example of symmetry-protected topological states. Haldane topological materials have potential applications in spintronics (i.e., study of electronic spin, in addition to its negative charge, in solid-state devices) and future quantum computers. We have recently shown [Ni(μ-4,4‘-bpy)(μ-ox)]n (NiBO; bpy = bipyridine) is a Haldane spin-1 chain material with a 2-D (2-dimensional or planar) MOF (metal-organic framework) structure. The work was published in Nature Communications and highlighted.
Figure 2. Ni2+ spin-1 Haldane chain showing the AF coupling (gray oval) between two unpaired electrons (red and blue arrows) in the middle of the chain, leaving unpaired electrons (orange arrows) at ends of the chain.
Selected Publications
Selected publications are listed below. To view a full list of publications, use this publication list.
Haldane Topological Spin-1 Chain in a Planar Metal-Organic Framework. Tin, P.; Jenkins, M. J.; Xing, J.; Caci, N.; Gai, Z.; Jin, R.; Wessel, S.; Krzystek, J.; Li, C.; Daemen, L. L.; Cheng, Y.; Xue, Z.-L., Nature Communications 2023, 14, 5454.
Comprehensive Studies of Magnetic Transitions and Spin-Phonon Couplings in the Tetrahedral Cobalt Complex Co(AsPh3)2I2. Moseley, D. H.; Liu, Z.; Bone, A. N.; Stavretis, S. E.; Singh, S. K.; Atanasov, M. Lu, Z.; Ozerov, M.; Thirunavukkuarasu, K.; Cheng, Y.; Daemen, L. L.; Lubert-Perquel, D.; Smirnov, D.; Neese, F.; Ramirez-Cuesta, A. J.; Hill, S.; Dunbar, K. R.; Xue, Z.-L., Inorganic Chemistry 2022, 61, 17123–17136.
Advanced Spectroscopic and Computational Studies of a Cobalt(II) Coordination Polymer with Single-Ion Magnet Properties. Tin, P.; Bone, A. N.; Bui, N. N.; Zhang, Y.-Q.; Chang, T.; Moseley, D. H.; Ozerov, M.; Krzystek, J.; Cheng, Y.; Daemen, L. L.; Wang, X.; Song, L.; Chen, Y.-S.; Shao, D.; Wang, X.-Y.; Chen, X.-T.; Xue, Z.-L. Journal of Physical Chemistry C. 2022, 31, 13268–13283.
Solution NMR of Transition Metal Complexes. Xue, Z.-L.; Cook, T. M., Invited review for the Comprehensive Inorganic Chemistry III, 2022. This review covers developments in solution NMR of compounds of 32 transition metals, including La, Lu and Ac, between 1990 and 2019.
Applying Unconventional Spectroscopies to the Single-Molecule Magnets, Co(PPh3)2X2 (X = Cl, Br, I): Unveiling Magnetic Transitions and Spin-Phonon Coupling. Bone, A. N.; Widener, C. N.; Moseley, D. H.; Liu, Z.; Lu, Z.; Cheng, Y.; Daemen, L. L.; Ozerov, M.; Telser, J.; Thirunavukkuarasu, K.; Smirnov, D.; Greer, S. M.; Hill, S.; Krzystek, J.; Holldack, K.; Aliabadi, A.; Schnegg, A.; Dunbar, K. R.; Xue, Z.-L., Chemistry – A European Journal 2021, 27, 11110-11125.
Inter-Kramers Transitions and Spin-Phonon Couplings in a Lanthanide-Based Single-Molecule Magnet. Moseley, D. H.; Stavretis, S. E.; Zhu, Z.; Guo, M.; Brown, C. M.; Ozerov, M.; Cheng, Y.; Daemen, L. L.; Richardson, R. ; Knight, G.; Thirunavukkuarasu, K.; Ramirez-Cuesta, A. J.; Tang, J.; Xue, Z.-L. Inorganic Chemistry 2020, 59, 5218-5230.
Neutron Instruments for Research in Coordination Chemistry. Xue, Z.-L.; Ramirez-Cuesta, A. J.; Brown, C. M.; Calder, S.; Cao, H.; Chakoumakos, B. C.; Daemen, L. L.; H., A.; Kolesnikov, A. I.; Mamontov, E.; Podlesnyak, A. A.; Wang, X. European Journal of Inorganic Chemistry 2019, 1065-1089.
Spin–Phonon Couplings in Transition Metal Complexes with Slow Magnetic Relaxation. Moseley, D. H.; Stavretis, S. E.; Thirunavukkuarasu, K.; Ozerov, M.; Cheng, Y.; Daemen, L. L.; Ludwig, J.; Lu, Z.; Smirnov, D.; Brown, C. M.; Pandey, A.; Ramirez-Cuesta, A. J.; Lamb, A. C.; Atanasov, M.; Bill, E.; Neese, F.; Xue, Z.-L. Nature Communications 2018, 9, 2572